Abstract
Introduction: Myeloablative regimens (MAC) including total body radiation (TBI) based conditioning regimes are commonly used in patients with acute leukemia undergoing hematopoietic stem cell transplant based on superior leukemia-free survival (Scott et al. 2017). MAC regimens are also associated with increased acute graft-versus-host disease (GVHD), a leading cause of non-relapse mortality. Hence, investigators have concentrated on controlling alloreactive immune activation resulting from TBI-induced tissue injury. A little is known about how stress-induced tissue damage and regeneration could independently increase tolerance against acute GVHD-associated inflammation, and alloreactive T-cell burden while achieving full donor bone marrow (BM) engraftment. To explore this, we used our recently developed 3D-image guided total marrow irradiation (TMI), which delivers targeted dose to the BM ensuring engraftment while reduces organ dose. We hypothesize, that reduced radiation dose on the Gastrointestinal (GI) tract using TMI will reduce GVHD by attenuating T cell trafficking to the GI without affecting donor chimerism.
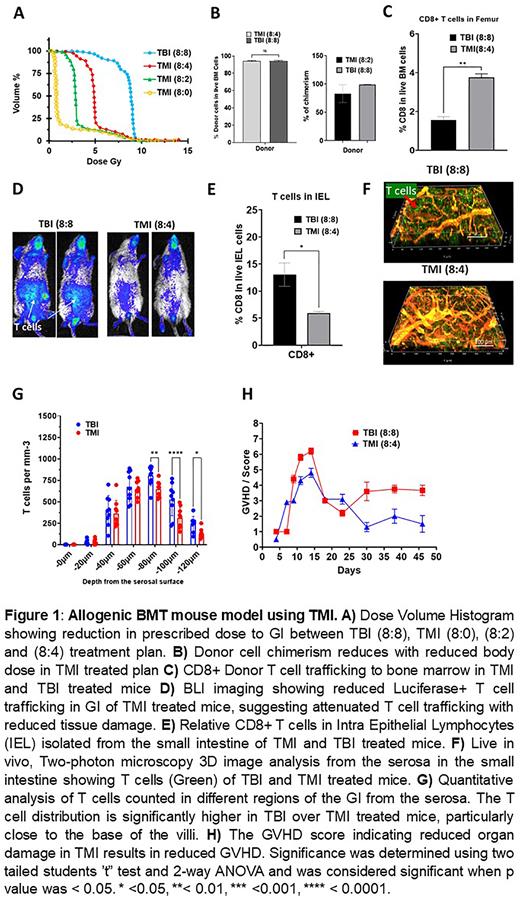
Method: The radiation treatment used in this study: TMI (8:0), TMI (8:2), TMI (8:4), and TBI (8:8), was developed as described (Darren et al., 2021). The dose to the organs were varied (0, 2, and 4 Gy), while the prescribed dose to the BM was 8 Gy. The host Balb/C (H2d) mice were irradiated in 2 fractions 6h apart and 24±1 h later were transplanted with 5 million T cell-depleted C57B/L 6 (H2b) donor BM cells with 1 million T cell enriched from donor spleenocytes. For bioluminescent imaging, Luciferase+ T cells from B6-Luc mice was used. Donor chimerism was assessed by anti-H2b and anti-H2d antibody staining by flow cytometry. The T cells were analyzed on day 7 post-BMT using flow cytometry. The GVHD scoring was carried out as described (Cooke et al.,1996). High-resolution multi-photon microscopy (MPM) was used to monitor trafficking of T cell and vascular network from Serosal side of the jejunum on Day 7 post-BMT and 3D image reconstruction was performed using Imaris software.
Results: While BM dose was kept at 8 Gy, the GI dose was reduced by 50% using TMI compared to TBI (Fig 1A). The donor engraftment was dependent on the body dose provided and required 4 Gy for complete engraftment as seen in TMI (8:4) treated mice while failed in TMI (8:0) or mixed-full engraftment observed in TMI (8:2) treated mice (Fig 1B). Histological analysis of the GI showed the damage was significantly reduced in TMI-treated mice in comparison to TBI-treated mice. The CD8+ T cells in the BM were significantly higher (Fig 1C), and consequently, the trafficking of CD8+ T cells to the GI was significantly reduced in TMI than in TBI treated mice (Fig 1D, E), suggesting differential targeting of Immune cells in TMI. Further, the 3D live MPM imaging of the GI shows an altered vascular niche in TBI relative to TMI-treated mice and correspondingly increased T cell numbers (Fig 1F). The T cells infiltration increased with increased dose to the GI as seen in TBI over TMI particularly from serosa surface towards the base of villi in the jejunum of the small intestine (Fig 1G). The consequences of reduced T cell trafficking and spatial T cell distribution in the GI of the TMI than TBI may have resulted in a reduced GVHD as assessed by scoring (Fig 1H).
Conclusion: The allogeneic immune cell reactivity that leads to GVHD could be directly a consequence of the increased trafficking of immune cells to the GI, arising at least partially from radiation-induced GI tissue injury during conditioning. However, this can be alleviated using our first-in-the-field preclinical TMI which ensures reduced GI dose, consequently reduced injury, and perhaps accelerated tissue repair. The in-vivo imaging suggests the T cell distribution varied depending on radiation dose and warrants future studies to understand the possibility of radiation dose dependent preferential niche for T cells in the GI and how this affects GVHD. Also, increasing BM targeted radiation could further increase the antileukemic effect, without increasing GI tissue injury. Therefore, future combinatorial trials with adoptive therapy could be further efficacious.
Disclosures
Salhotra:BMS: Research Funding; Orca Bio: Research Funding; Kadmon: Other: Advisory board meeting . Hui:Janssen Research & Development, LLC.: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal